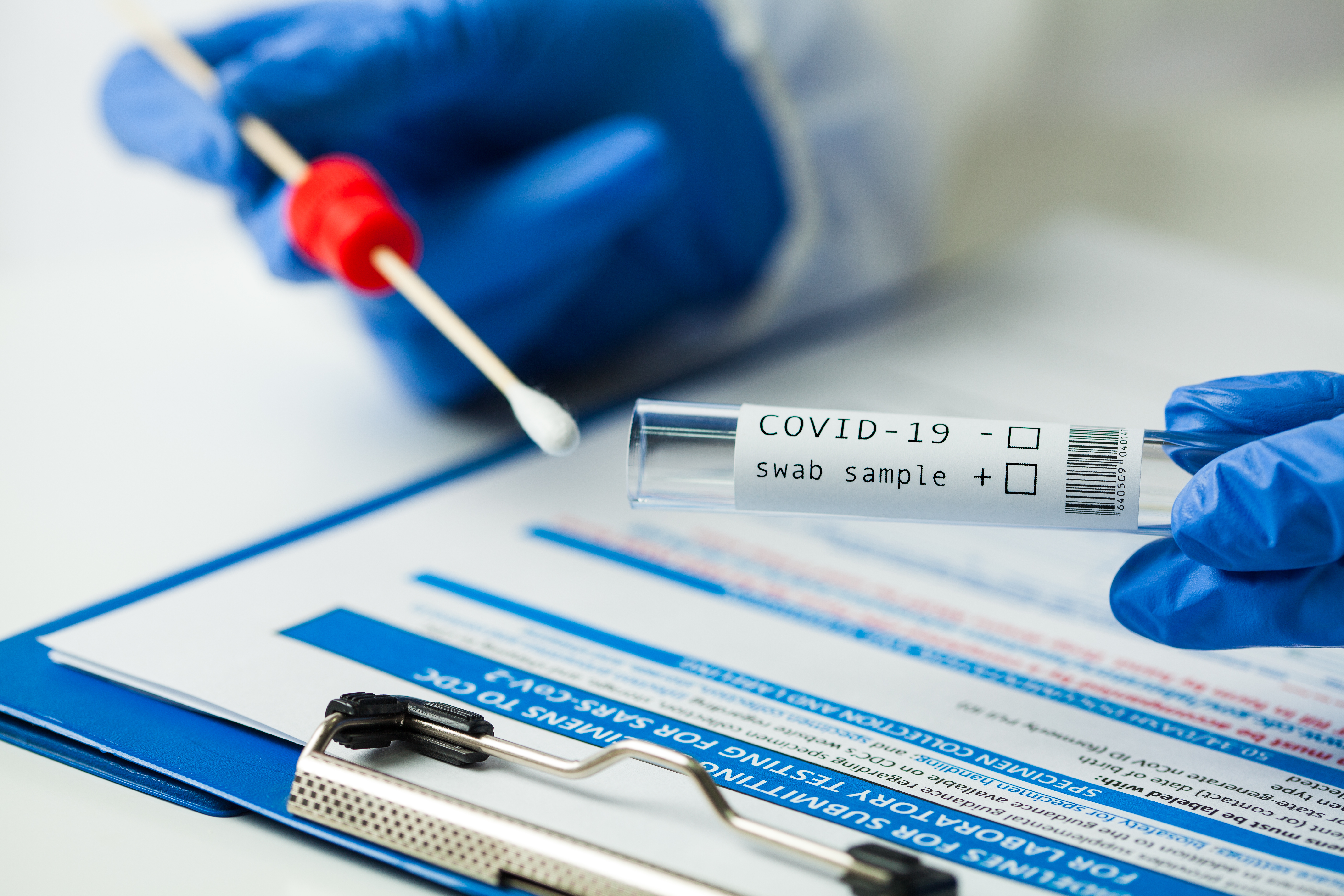
Eurofins U.S. Clinical Diagnostics announced the launch date of its pooled PCR test to detect SARS-CoV-2 and significantly reduce the price per PCR test for clients. Pooling tests, historically used in blood banks, can now be used by the myriad of industries seeking a highly accurate and cost-effective means to conduct COVID-19 testing. Those industries include corporations, sports organizations, educational institutions, congregate care/nursing, and manufacturing.
Eurofins Launches Lower Cost Highly Accurate COVID-19 PCR Test
Tags: CRO
For the 2020 CRO Leadership Awards, Life Science Leader magazine once again teamed up with Industry Standard Research (ISR) to determine the award recipients. More than 60 contract research organizations were assessed on over 20 performance metrics in ISR's annual CRO Quality Benchmarking survey. Eurofins is recognized as a recipient of these awards, exceeding customer expectations in Quality, Capabilities and Compatibility.
Tags: CRO
When Choosing a Central Lab, No One Gets Fired for Choosing Quality!
For those that have been in the industry for awhile, we have all heard the saying "No one gets fired for choosing "Company X"! You can insert the company name of your choice. In my 30 years in clinical research, I've heard this in reference to a couple of companies.
The question that needs to be asked is, are you and your organization making this statement based upon:
- Consistent commitment to quality and compatibility?
- Extensive global capabilities?
- Reliable service year after year?
Or is it being made due to:
- Inertia?
- Historical relationship – better to stick with the devil you know?
- Understaffed/overworked vendor management personnel, challenged with finding best in class in solution?
These are very real phenomena that I'm sure we have all experienced at one time or another. But if you are making the statement due to the latter, they all indicate that your clinical development team may be missing out on receiving Best in Class central laboratory solutions.
When choosing a central laboratory, here are a few factors you should look for:
- Quality Systems
- Global Standardization
- Logistics Services
- Data Management and IT Services
- Additional Services
At Eurofins, we provide seamless, end-to-end solutions to help clients progress through the drug development cycle through a single, experienced provider. From compound discovery and clinical research through the manufacture and release of commercial product and post-approval/marketing, our integrated solutions deliver the most comprehensive range of services. Our state-of-the-art analytical technologies and expansive geographic reach enable us to support our clients’ specialized testing needs and stringent quality and safety requirements.
And, with Eurofins being recognized for the 7th year in a row – every year since the inception – of the CRO Leadership Award (1 of only 2 laboratory providers able to state that fact), shouldn't the statement read:
Tags: CRO