Tracy Hendershott
As Vice President of Commercial Operations at Eurofins Central Laboratory, Tracy Hendershott has three decades of ever increasing responsibility in the Clinical Trial arena, both in the CRO and Central Laboratory fields.
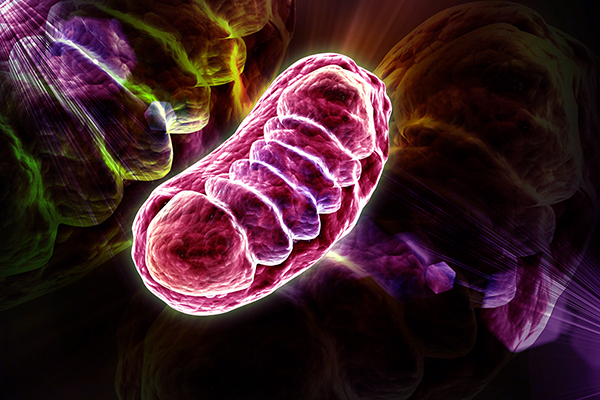
Primary Mitochondrial Myopathy (PMM)
Rare diseases are one of the most challenging specialties within clinical trials, due to limited patient populations, lack of historic large scale studies to draw knowledge/clinician expertise and build upon, and frequently, lack of necessary funding to conduct thorough and multi-phased research. That is why Eurofins Central Laboratory is delighted to once again participate with Orphan Reach, Emmes’ rare disease CRO, and their extensive expertise with overcoming these challenges.
Read More
Eurofins Central Laboratory is pleased to collaborate with The Emmes Company in the conduct of Diabetic Foot Osteomyelitis (DFO) clinical trials.
Read More
While we have numerous symptomatic treatments today, there is still no cure for neurodegenerative diseases such as Parkinson’s and Alzheimer’s. There are more than 600 neurodegenerative diseases alone that affect the brain, spine, and peripheral nervous system. While diseases like Parkinson’s affect motor functions, Alzheimer’s is a progressive disease that destroys memory and other mental functions.
Read More
Tags:
Central Laboratory,
BioPharma,
Alzheimer's Disease,
Neurodegenerative,
Parkinson's Disease
At Eurofins BioPharma Services, we feel this FDA press release, as a Christmas gift that came early! After working on multiple protocols, across multiple clients, we couldn't be happier to see the continued evolution of treatment solutions for this rare disease (around 2 cases per million). Prior to the first treatment being released in 2007, life expectancy of patient diagnosed with PNH was only ten years, with poor quality of life throughout that time. With the significant resource dedication of years of effort and capital, our peers and colleagues throughout the clinical trial industry have allowed patients to have:
Read More
Tags:
FDA,
Central Laboratory,
Clinical Trial,
PNH,
New Treatment
We would like to congratulate our colleagues at Eurofins DiscoverX for their recent patent application approval for improvements to compound screening and evaluation on cellular responses.
Read More
Tags:
Cellular Response,
DiscoverX,
Compound Screening
Currently, there aren't any globally-approved pharmaceutical treatments for Stress Urinary Incontinence (SUI). With a predominately female patient population ranging in the hundreds of millions of people worldwide, this provides them with only surgical and physical therapy-based options, limiting their choice of solutions. While effective for some, there can be challenges that come with the invasive nature of surgical procedures, as well as non-compliance with the length of time required for successful physical therapy outcomes.
Read More
Tags:
Pharmaceutical,
BioPharma,
Stress Urinary Incontinence
Testicular Toxicity
After being in the draft stages for approximately three years and continuing to receive industry feedback, the FDA CDER has just released final Guidance for Industry Title Testicular Toxicity: Evaluation During Drug Development in October 2018. There are numerous challenges with conducting multi-center clinical trials focusing on testicular toxicity and yet remaining within the realm of guidance from the FDA per their statement:
Read More
Tags:
FDA,
Central Laboratory,
Testicular Toxicity,
Clinical Trial
In the realm of infectious disease, antibiotic resistance is a real and ever increasing concern across the entire healthcare industry. This is also true for ocular infections. With bacterial conjunctivitis being the second most common cause (versus viral conjunctivitis), diligent epidemiology and surveillance monitoring brings valued insight to us all. Eurofins Central Laboratory is proud to have provided analytical services (microbiological species confirmation and antibiotic resistance profile determination) to one of the only two nationwide multi-center surveillance studies (Antibiotic Resistance Monitoring in Ocular Microorganisms - ARMOR) ever undertaken in this indication. Article can be downloaded here.
Read More
Tags:
Clinical Trial,
Drug Development,
Bioanalytical Testing
World Chronic Obstructive Pulmonary Disease (COPD) Day is organized by the Gloobal Initiative for Chronic Obstructive Lung Disease (GOLD) in collaboration with health care professionals and COPD patient groups throughout the world. World COPD Day is celebrated on November 15th, 2018, and it's aim is to raise awareness about the disease and improve outcomes throughout the world.
Read More
Tags:
Central Laboratory,
Clinical Trial,
Drug Development